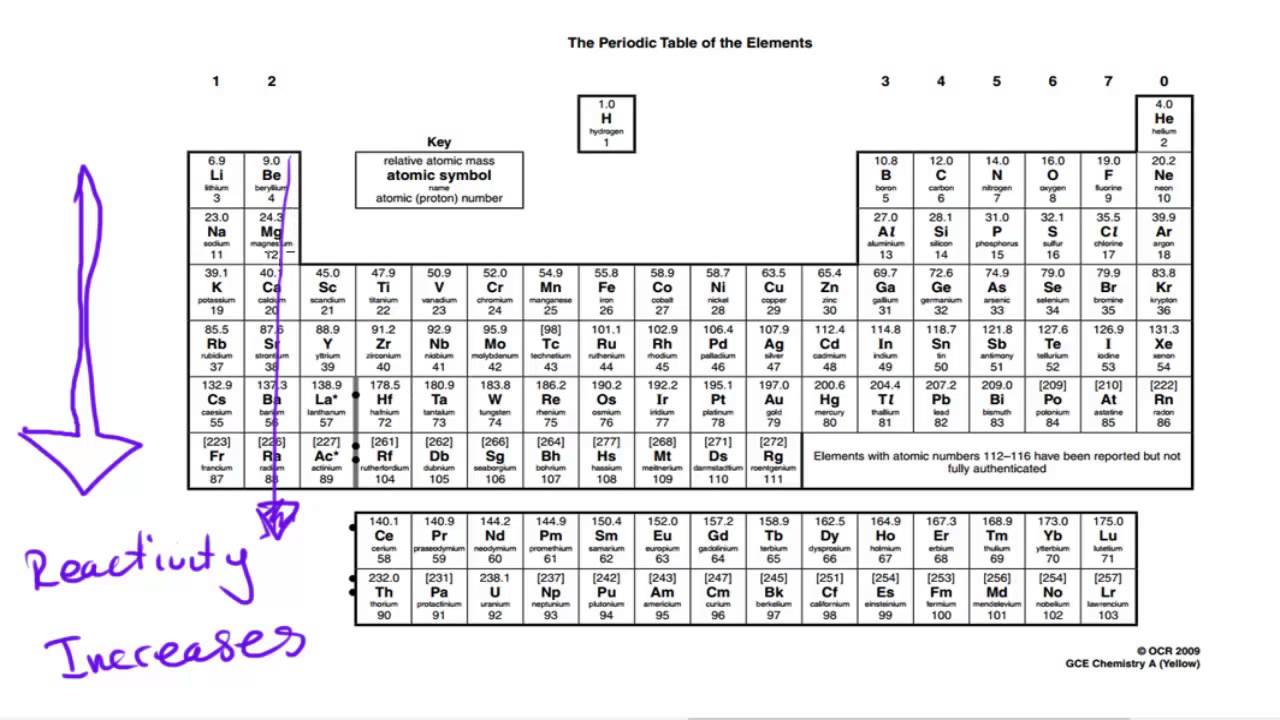
Properties of Metals Metals are good conductors of heat and electricity. These are the electrons that are transferred or shared when atoms bond together. Valence electrons are the electrons in the outer energy level of an atom. Valence Electrons The number of valence electrons an atom has may also appear in a square. It can consist of a single capital letter, or a capital letter and one or two lower case letters. Symbols All elements have their own unique symbol. An atomic mass number with a decimal is the total of the number of protons plus the average number of neutrons. Some atoms have more or less neutrons than protons. What is its atomic number?Ītomic Mass and Isotopes While most atoms have the same number of protons and neutrons, some don’t. Its atomic mass is 4 (protons plus neutrons). It is derived at by add ing the number of protons with the number of neutrons. Bohr Model of Hydrogen Atom Wave ModelĪtomic Mass Atomic Mass refers to the “weight” of the atom. No two elements, have the same number of protons. Ītomic Number This refers to how many protons an atom of that element has. What’s in a square? Different periodic tables can include various bits of information, but usually: atomic number symbol atomic mass number of valence electrons state of matter at room temperature. No two elements have the same atomic number. The atomic number is unique to that element. For instance, hydrogen has 1 proton, so it’s atomic number is 1.  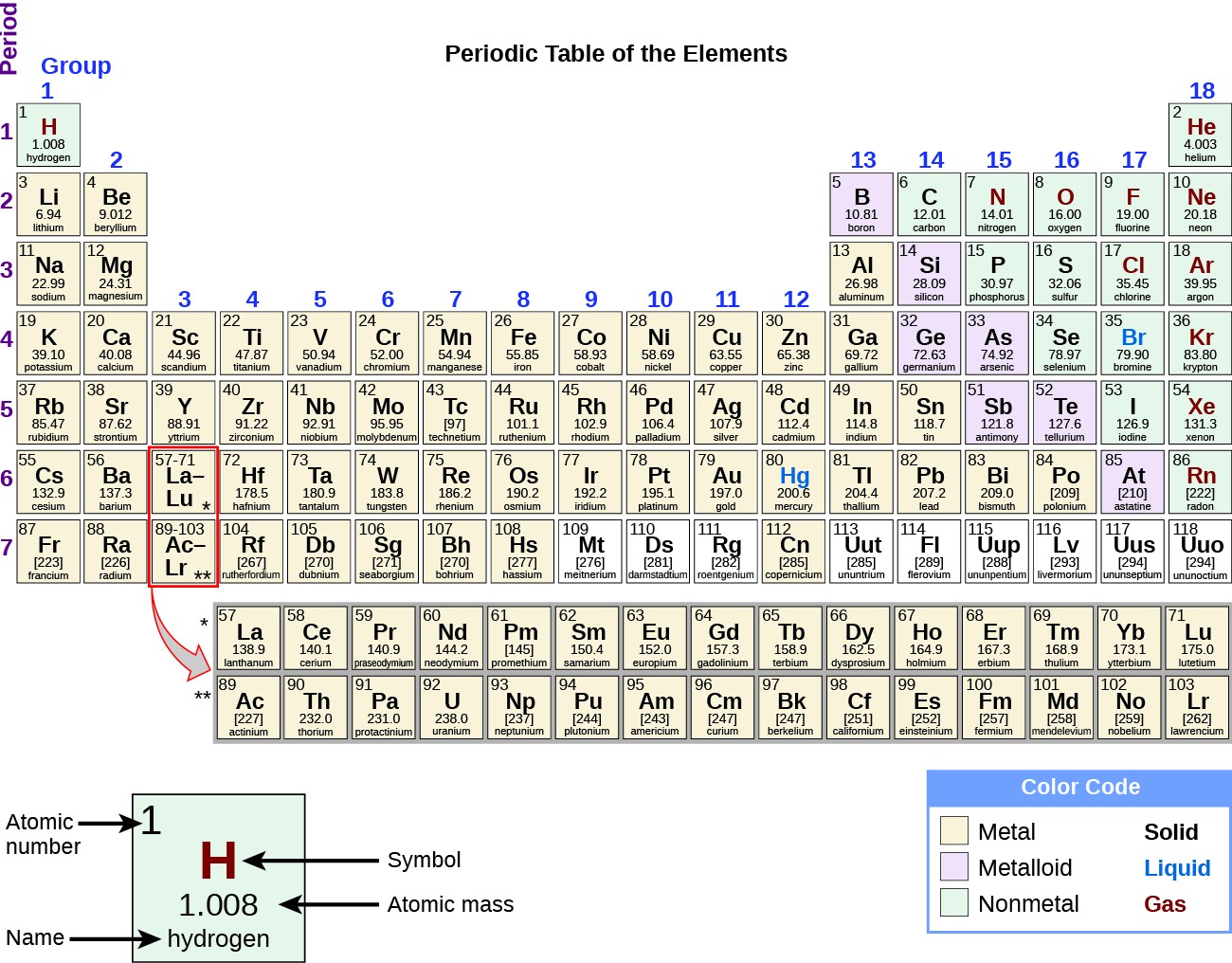
The atomic number refers to how many protons an atom of that element has. Key to the Periodic Table Elements are organized on the table according to their atomic number, usually found near the top of the square. Understanding the organization and plan of the periodic table will help you obtain basic information about each of the 118 known elements. You can also predict how an element will react chemically with another element. For example, you can predict with reasonably good accuracy the physical and chemical properties of the element. A great deal of information about an element can be gathered from its position in the period table. Periodic Table The periodic table organizes the elements in a particular way. The most abundant element in the earth’s crust is oxygen. Įlements The elements, alone or in combinations, make up our bodies, our world, our sun, and in fact, the entire universe. Scientists have identified 90 naturally occurring elements, and created about 28 others. (The above picture of the periodic system is interactive - no need to download, just click on an element.Gold silver helium oxygen mercury hydrogen sodium nitrogen niobium neodymium chlorine carbonĮlements Science has come along way since Aristotle’s theory of Air, Water, Fire, and Earth. Now available: history of the periodic tableĬhoose elements by name, by atomic number, by symbol, by massĬlick here for the history of the periodic table.Ĭlick here to download a PDF version from that periodic table An interactive, printable extended version of the Periodic table of chemical elements of Mendeleev (who invented the periodic table). Phone: +31 152 610 900 chemical element contains a link to a page that explains its chemical properties, health effects, environmental effects, application data, an image and also information of the history/inventor of each element.Separation and Concentration Purification Request. 
Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
